
Starting from the bottoms, point ( x xB 0.02, y x B), a bubble point calculation with x 0. The Kellogg and DePriester charts and their subsequent extensions and generalizations use the molar average boiling points of the liquid and vapor phases to represent the composition effect. Vapor pressure for the pure species are given by the following Antoine equations: i ii a)Prepare a graph showing P vs. 4-38 Table 4.4-6 lists the Matlab codes for the number of equilibrium stages. SI versions of these charts have been developed by Dadyburjor. D20 Use the DePriester chart to generate the temperature-composition. For K as a function of T and P only, the DePriester charts provide good starting values for the iteration. One cannot calculate K values until phase compositions are known, and those cannot be known until the K values are available to calculate them. The Kellogg charts, and hence the DePriester charts, are based primarily on the Benedict-Webb-Rubin equation of state, which can represent both the liquid and the vapor phases and can predict K values quite accurately when the equation constants are available for the components in question.Ī trial-and-error procedure is required with any K-value correlation that takes into account the effect of composition. source of K-values for light hydrocarbons is the well-known De Priester charts. These charts are a simplification of the Kellogg charts and include additional experimental data. complete graph for the set of equations is shown in Figure 1.10. The easiest to use are the DePriester charts, which cover 12 hydrocarbons (methane, ethylene, ethane, propylene, propane, isobutane, isobutylene, n-butane, isopentane, n-pentane, n-hexane, and n-heptane). Other names: Hexane Skellysolve B n-C6H14 Esani Heksan Hexanen Hexyl hydride Gettysolve-B NCI-C6052. For example, several major graphical K-value correlations are available for light-hydrocarbon systems. The 3d structure may be viewed using Java or Javascript.
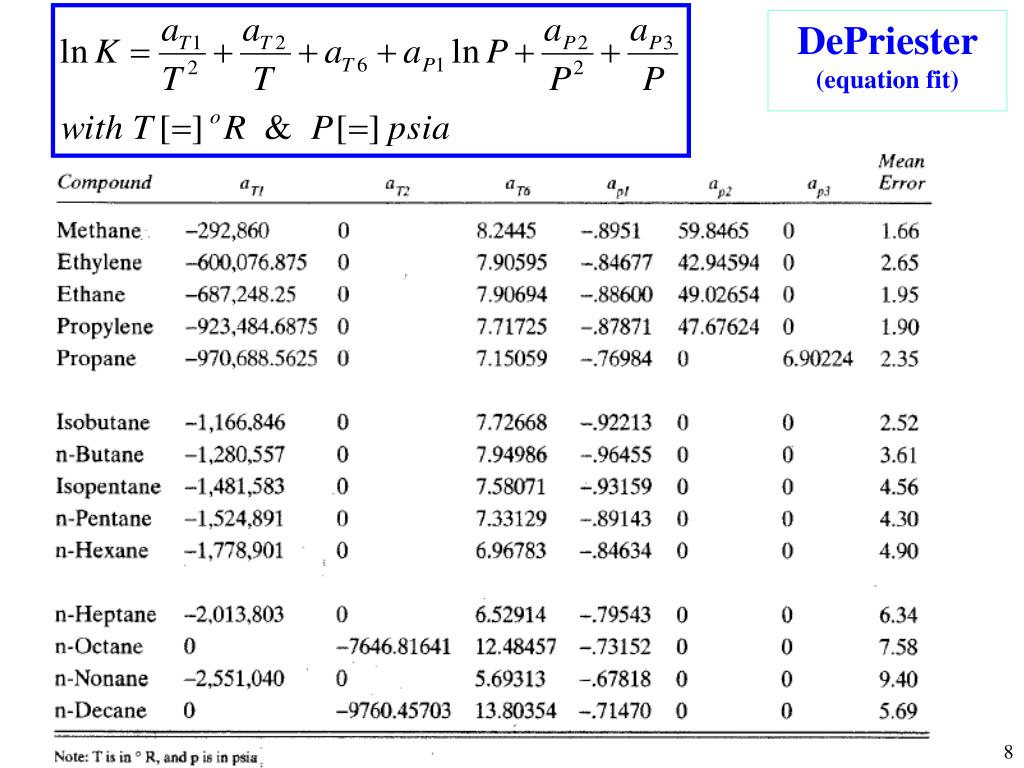
(2) We can use K-values directly, by looking values up (DePriester Charts) or using correlations. However, for mixtures of compounds of similar molecular structure and size, the K value depends mainly on temperature and pressure. and Henrys Law expression as: K y i /x i H i (T)/P. 4, the K value of a species is a complex function of temperature, pressure, and equilibrium vapor- and liquid-phase compositions.
